Glucagon-like peptide-1 (GLP-1) medications have conquered the weight loss market.
Currently, about one in eight American adults are taking GLP-1 drugs but despite their effectiveness and popularity, they cause side effects, are expensive, and artificial. And they often leave patients without guidance when they have reached their weight loss goals and are ready to stop using the prescription drugs but don’t want the weight coming back on.

What is Glucagon-Like Peptide-1?
GLP-1 is a hormone that plays a key role in controlling appetite, blood sugar, and energy balance. It is produced in the gut by specific bacteria.
Enter Akkermansia muciniphila, a friendly gut bacterium, that naturally supports GLP-1 production. Since it was discovered in 2004, Akkermansia muciniphila has become one of the most talked-about microbes in gut health research. Low levels of Akkermansia in the gut have been linked to obesity, insulin resistance, and metabolic syndrome, while increased levels are associated with lean body types and improved glucose control.
The link between Akkermansia and GLP-1 offers easy to implement and affordable ways for supporting natural metabolic balance. By stimulating GLP-1 production and strengthening the gut barrier, Akkermansia can help:
- Improve insulin sensitivity and blood sugar control
- Keep you feeling fuller for longer, reducing food intake
- Lower inflammation that’s associated with metabolic dysfunction
- Support a healthy gut lining that’s crucial for immune balance and nutrient absorption.
Whereas other gut microbes break down dietary fiber for sustenance, Akkermansia feeds on mucin in the lining of the gut, triggering increased mucin production and preventing leaky gut syndrome. You can think of it like using peeling on your skin to encourage skin cell renewal.
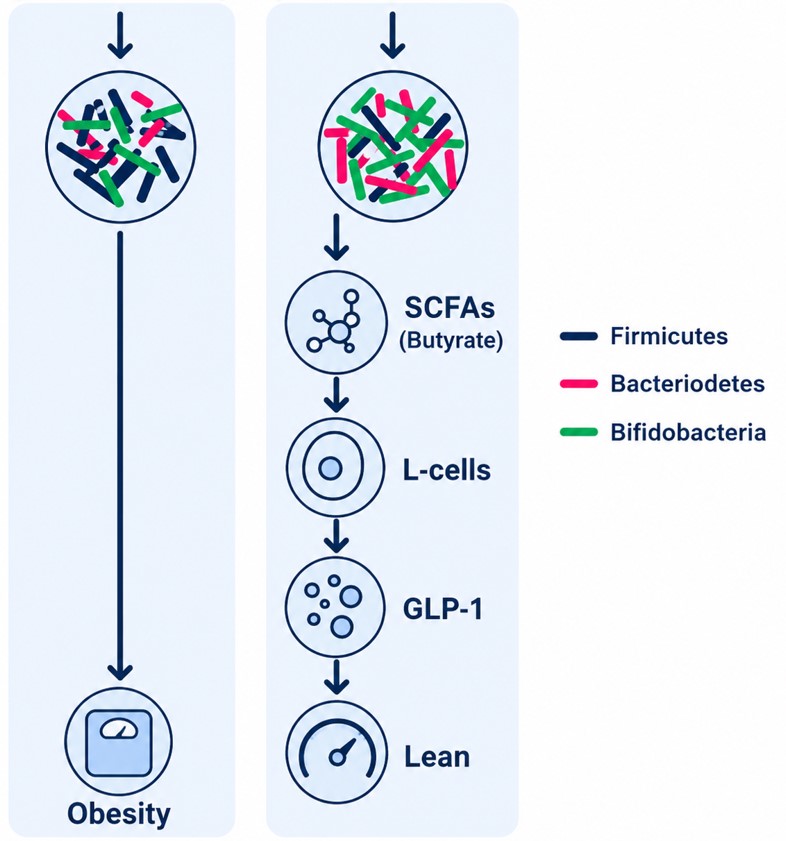
There are other beneficial gut bacteria that work synergistically. Probiotics that increase GLP-1 have in common that they work by enhancing short-chain fatty acid (SCFA) production. Akkermansia generates short-chain fatty acids (SCFAs) such as acetate and propionate by breaking down mucin. Clostridium butyricum, another friendly gut bacterium, produces butyrate. SCFAs such as acetate, propionate, and butyrate bind to L-cells in the intestinal lining stimulating the release of GLP-1 while lowering inflammatory biomarkers.

Top Probiotics and Strains for GLP-1 Support
- Akkermansia muciniphila: Known as a key bacterium for gut lining health SCFAs (acetate and propionate) and also a specialized protein (P9) along with smaller amounts of branched-chain fatty acids (BCFAs) and succinate, and stimulates gut L-cells to release more GLP-1.
- Clostridium butyricum: Produces butyrate, providing energy to gut cells and stimulating the release of GLP-1. The combination of C. butyricum and A. muciniphila (CB-AKK) significantly strengthens the intestinal mucosal barrier, reduces leaky gut, and lowers inflammation (lower IL-1β, IL-6, and TNF-α).
- Bifidobacterium and Lactobacillus strains: Including B. infantis, L. plantarum, and L. acidophilus.
- Lactobacillus kefiranofaciens M and Lactobacillus kefiri K: Identified for their ability to enhance GLP-1 secretion.
Akkermansia strengthens cancer prevention
Recent studies have found that Akkermansia in combination with Lactobacillus infantis and Clostridium play a significant role in activating the immune system, reshaping the tumor microenvironment, and inhibiting cancer progression. The strongest protective effects was shown for breast cancer and colitis-associated colorectal cancer prevention.
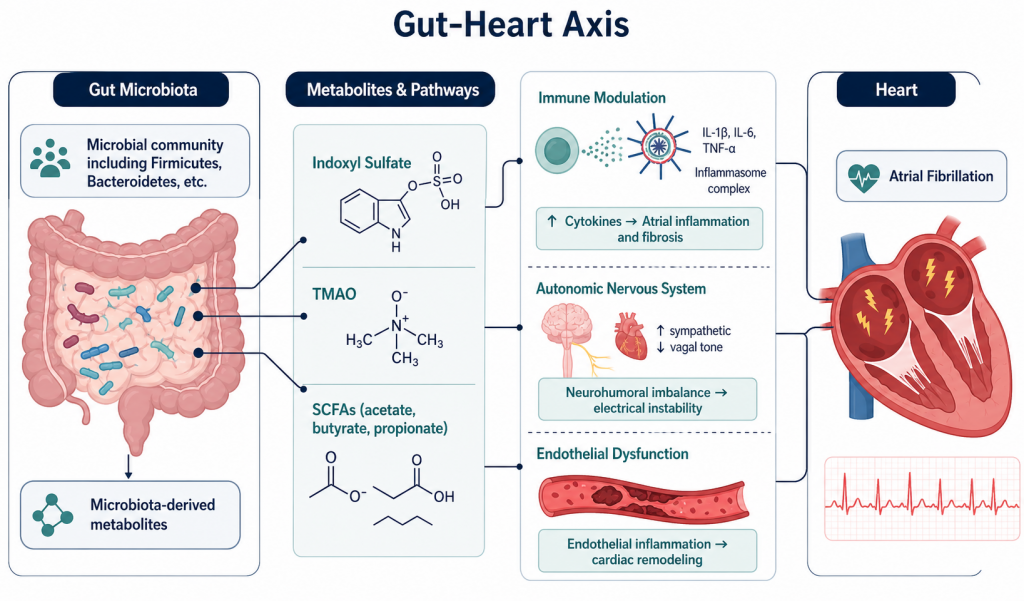
Gut dysbiosis and Atrial fibrillation (A fib)
Patients with A Fib have altered gut microbiota compared with healthy individuals, with changes in specific bacterial groups. Low levels of Akkermansia muciniphila and a reduced abundance of certain Firmicutes bacteria are strongly associated with gut dysbiosis, especially when there are low levels of Firmicutes and high levels of Bacteroidetes. Gut dysbiosis contributes to atrial fibrillation onset by producing harmful metabolites such as trimethylamine N-oxide and indoxyl sulfate, reducing protective short-chain fatty acids, and triggering systemic inflammation, myocardial fibrosis, and electrical changes in the heart. Such state of microbial imbalance can increase intestinal permeability—or “leaky gut”—allowing inflammatory toxins like lipopolysaccharides (LPS) into the bloodstream.
Metabolic syndrome, including obesity, diabetes, and dyslipidemia, elevates the risk of A Fib by many mechanisms, including inflammation. In addition, lifestyle factors such as excessive alcohol consumption, low physical activity, and sleep disorders such as obstructive sleep apnea are modifiable contributors to AF development and progression.
In return, AF can worsen gut health by causing low blood flow to the intestines, increasing gut permeability, and allowing bacterial toxins into circulation, which further fuels inflammation.
Targeting the gut microbiota through diet and probiotics may reduce inflammation and atrial remodeling, offering a new way to manage AF.
Nourish Your Gut Microbiome during GLP-1 Therapy and Longterm
Cut back on processed foods, greasy and fried foods, added sugars, and alcoholic beverages. This way you take food away from harmful bacteria and help keep inflammation at bay. Enjoy foods high in fiber, like fruits, vegetables, leafy greens, beans, whole grains such as farro and quinoa, nuts, and seeds to feed beneficial gut bacteria. For proteins prioritize whole foods such as wild-caught fish, organic tofu, organic chicken, chia seeds, and beans over processed meats. Fermented foods, such as yogurt, kefir, kombucha, sauerkraut, and kimchi, add good bacteria and maintain a balanced diversity in your gut microbiome.
A Holistic Approach to GLP-1 Wellness
Quality sleep plays a crucial role in digestive health and long-term weight management.
Staying properly hydrated supports digestive function and can help minimize some GLP-1 side effects.
Keeping your muscles strong while losing weight can support day-to-day functions and overall wellness. Making sure your body gets enough amino acids and minerals is crucial to maintain muscle tissue.
At MANYA, we offer Pendulum Professional-Grade Probiotics and Polyphenols. The Pendulum multi-strain formula combines Akkermansia, Clostridium butyricum, and Bifidobacterium infantis to enhance natural GLP-1 production and curb cravings while amplifying your body’s fullness signal naturally. Professional-grade Akkermansia delivers 5x higher potency than consumer versions, ensuring robust microbiome replenishment. Schedule your appointment today to receive guidance for lifestyle changes. We also offer comprehensive functional lab test including GI Map stool map and mold panels.

Disclaimer:
The information provided in this blog post is for informational purposes only and is not intended as medical advice. The content shared here is based on general wellness principles and is not a substitute for professional medical evaluation, diagnosis, or treatment. This blog does not establish a patient-provider relationship, and the opinions expressed are solely those of the author. Please seek the guidance of your physician or another qualified healthcare professional for any medical concerns you may have including but not limited to diabetes and Afib. Always consult with your healthcare provider before starting any new supplement regimen or making changes to your current health plan. Individual results may vary. The products in this article are not intended to diagnose, treat, cure, or prevent any disease.
Sources:
Abdalqadir N, and Khosrow A. GLP-1 and GLP-2 Orchestrate Intestine Integrity, Gut Microbiota, and Immune System Crosstalk, Microorganisms, Vol. 10, Issue 10, 19 Oct. 2022, article 2061. .
Awashra A, Neiroukh H, et al. The gut–heart axis in atrial fibrillation: Pathophysiology, evidence, and therapeutic potential, Heart Rhythm O2, Vol. 7, Issue 3, 2026, Pages 581-597, https://doi.org/10.1016/j.hroo.2025.10.013.
Arukha AP, Nayak S, Swain DM. Effect of Akkermansia muciniphila on GLP-1 and Insulin Secretion. Nutrients. Vol. 17, Issue 15, 31 Jul 2025, p. 2516, https://doi.org/10.3390/nu17152516.
Cani, PD, and Knauf C. A Newly Identified Protein from Akkermansia muciniphila Stimulates GLP-1 Secretion. Cell Metabolism, vol. 33, no. 6, 1 June 2021, pp. 1073–1075. Elsevier, https://doi.org/10.1016/j.cmet.2021.05.004.
Chen J, Wang Y, et al. Exploring the Axis of Gut Microbiota-Inflammatory Cytokine-Atrial Fibrillation in the Pathogenesis of Atrial Fibrillation. J Cell Mol Med, Vol. 29, Issue 3, Feb 2025, https://doi.org/10.1111/jcmm.70379.
Cronin, Peter, et al. “Dietary Fibre Modulates the Gut Microbiota.” Nutrients, vol. 13, no. 5, 13 May 2021, article 1655. PubMed, https://doi.org/10.3390/nu13051655.
Duncan, Jessica, et al. “Individualized Virtual Integrative Medicine (IVIM): A Clinical Model for Enhanced GLP-1 Therapeutic Outcomes.” Obesity Pillars, vol. 15, 30 June 2025, article 100188. https://pmc.ncbi.nlm.nih.gov/articles/PMC12272120/.
Gofron, Krzysztof Ksawery, Andrzej Wasilewski, and Sylwia Małgorzewicz. Effects of GLP-1 Analogues and Agonists on the Gut Microbiota: A Systematic Review. Nutrients, Vol. 17, Issue 8, 2025, article 1303, https://doi.org/10.3390/nu17081303.
Here, Yoon et al. (2021) have identified a novel protein (P9) secreted by A. muciniphila that stimulates GLP-1 secretion, thereby adding new insight to the biomolecule era to treat metabolic diseases.
Hua D, Yang Q, et al. The combination of Clostridium butyricum and Akkermansia muciniphila mitigates DSS-induced colitis and attenuates colitis-associated tumorigenesis by modulating gut microbiota and reducing CD8+ T cells in mice. mSystems. Vol. 10, Issue 2, 2025 Feb 18, https://doi.org/10.1128/msystems.01567-24.
Jalleh RJ, Rayner CK, et al. Gastrointestinal Effects of GLP-1 Receptor Agonists: Mechanisms, Management, and Future Directions. The Lancet Gastroenterology & Hepatology, Vol. 9, Issue 10, Oct. 2024, pp. 957–964. ScienceDirect, https://doi.org/10.1016/S2468-1253(24)00188-2.
Kolterman O, Perlman A. Harnessing GLP-1 in the Digestive Tract for Better Metabolic Health. Pendulum Therapeutics; 2024. Accessed June 16, 2025. https://cdn.shopify.com/s/files/1/0175/3104/3894/files/Harnessing-GLP-1-White-Paper-v2-May-2024-web.pdf
Li X, Hua D, et al. Oral Combined Probiotics Clostridium butyricum and Akkermansia muciniphila Inhibits the Progression of 4T1 Breast Cancer by Activating Bcl-2/Bax Pathway. Cancer Medicine, Vol. 14, Issue 11, https://doi.org/10.1002/cam4.70987.
Mohseni, Saeid, et al. Curcumin for the Clinical Treatment of Inflammatory Bowel Diseases: A Systematic Review and Meta-Analysis of Placebo-Controlled Randomized Clinical Trials. Frontiers in Nutrition, Vol. 12, Issue 23 Mar. 2025, https://doi.org/10.3389/fnut.2025.1494351.
Cani PC, Knauf C. A newly identified protein from Akkermansia muciniphila stimulates GLP-1 secretion, Cell Metabolism, Volume 33, Issue 6, 2021, Pages 1073-1075, https://doi.org/10.1016/j.cmet.2021.05.004.
Pendulum Therapeutics. [Clinical Applications of Akkermansia muciniphila and Probiotic Synergy]. 2024. Accessed June 16, 2025. https://cdn.shopify.com/s/files/1/0175/3104/3894/files/CLIN.25_PMD_Ovation_Labs_White_Paper_v.pdf
Plovier H, Everard A, et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat Med. Vol. 23, Issue 1, pages 107-113, Jan 2017, https://doi.org/10.1038/nm.4236.
Rodrigues, Fernandes V, et al. Akkermansia Muciniphila and Gut Immune System: A Good Friendship That Attenuates Inflammatory Bowel Disease, Obesity, and Diabetes. Frontiers in Immunology, Vol. 13, Article 934695, 7 July 2022, https://doi.org/10.3389/fimmu.2022.934695.
Rodríguez-Daza MC, de Vos WM. Polyphenols as Drivers of a Homeostatic Gut Microecology and Immuno-Metabolic Traits of Akkermansia muciniphila: From Mouse to Man. International Journal of Molecular Sciences. Vol. 24, Issue 1, p. 45, 2023, https://doi.org/10.3390/ijms24010045
Sheng, Wei, Guang Ji, and Li Zhang. Immunomodulatory Effects of Inulin and Its Intestinal Metabolites. Frontiers in Immunology, Vol. 14, 2023, Article 1224092, https://doi.org/10.3389/fimmu.2023.1224092.
Sun, Jingyi, et al. Sleep Deprivation and Gut Microbiota Dysbiosis: Current Understandings and Implications. International Journal of Molecular Sciences, Vol. 24, Issue 11, 31 May 2023, Article 9603, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10253795/.
Thomas, Liji. GLP-1 Agonists May Reshape the Gut Microbiome. News-Medical.net, 11 Apr. 2025, https://www.news-medical.net/news/20250411/GLP-1-agonists-may-reshape-the-gut-microbiome.aspx.
Tsai, Chih-Yiu, et al. Gut Microbial Signatures for Glycemic Responses of GLP-1 Receptor Agonists in Type 2 Diabetic Patients: A Pilot Study, Frontiers in Endocrinology, Vol. 12, Article 814770, 10 Jan. 2022. Frontiers, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8793908/.
Zeng X, et al. Gut Bacterial Nutrient Preferences Quantified in Vivo, Cell, Vol. 185, Issue 18, 1 Sept. 2022, pp. 3441–3456.e19. Elsevier, https://doi.org/10.1016/j.cell.2022.07.020.
Zeng Y, Wu Y, et al. Crosstalk between Glucagon-Like Peptide 1 and Gut Microbiota in Metabolic Diseases, mBio, Vol. 15, Issue 1, 6 Dec. 2023, https://doi.org/10.1128/mbio.02032-23.
Zhou, QiQi, et al. Randomised Placebo-Controlled Trial of Dietary Glutamine Supplements for Postinfectious Irritable Bowel Syndrome. Gut, Vol. 68, Issue 6, June 2019, pp. 996–1002. BMJ Publishing Group, https://doi.org/10.1136/gutjnl-2017-315136.




